A small RNA drug delivered broad lipid-lowering signals in an early human study
An early-stage clinical trial has added momentum to a new approach for treating difficult lipid disorders. In a final phase 1 report published in Nature Medicine, researchers said the investigational therapy zodasiran lowered triglycerides and reduced circulating angiopoietin-like protein 3, or ANGPTL3, in patients already receiving lipid-lowering treatment. The study included people with hyperlipidemia, familial hypercholesterolemia, and moderate-to-severe hypertriglyceridemia, offering an early look at whether one molecular target could help across several forms of dyslipidemia.
Zodasiran is a small interfering RNA designed to target ANGPTL3, a protein linked to triglyceride and low-density lipoprotein cholesterol regulation. The rationale behind the drug is grounded in human genetics. The paper notes that loss-of-function variants in the gene encoding ANGPTL3 are associated with lower triglyceride and LDL cholesterol levels as well as lower cardiovascular risk. That makes ANGPTL3 an attractive target for a medicine intended to produce durable lipid changes through intermittent dosing.
What the phase 1 trial tested
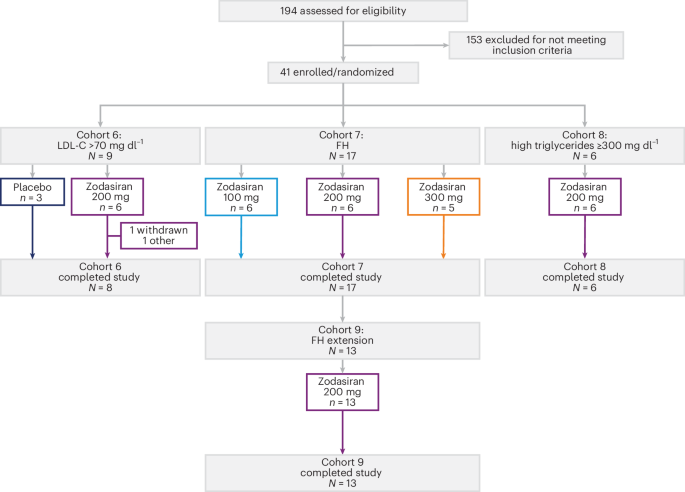
The trial ran for 16 weeks and enrolled patients on background lipid-lowering therapy in three groups: hyperlipidemia, familial hypercholesterolemia, and moderate-to-severe hypertriglyceridemia. The hyperlipidemia cohort included a placebo control arm. In total, 32 patients were described in the main study population: 9 with hyperlipidemia, 17 with familial hypercholesterolemia, and 6 with moderate-to-severe hypertriglyceridemia.
Participants received zodasiran by subcutaneous injection on day 1 and day 29. The study also included a 48-week open-label extension in the familial hypercholesterolemia cohort, where zodasiran was given every 12 weeks. That extension was important because it offered a first read on whether suppressing ANGPTL3 could remain durable beyond the initial treatment window.
The primary endpoint was safety, specifically the occurrence of serious treatment-related adverse events. On that measure, the study produced an encouraging result for the program. Investigators reported that no serious treatment-related adverse events were observed. They also reported no elevations in hepatic aminotransferases, bilirubin, or glycated hemoglobin, and no drug discontinuations.
Magnitude of the lipid effects
The secondary endpoint results were strong enough to make this study notable despite its small size. At week 16, which was 12 weeks after the second dose, all cohorts showed reductions in serum ANGPTL3 of up to 85.4% and triglyceride reductions of up to 67.1%. The paper said ANGPTL3 reduction was sustained through the end of the open-label extension in the familial hypercholesterolemia cohort.
That matters because the study was not focused on a single narrow patient population. Familial hypercholesterolemia and severe triglyceride disorders present different clinical problems, and patients are often already using other lipid-lowering drugs. A signal across these groups suggests the mechanism may have wider utility than a therapy aimed at only one biomarker in one disease setting.
The study does not establish cardiovascular outcomes, and it does not answer where zodasiran would ultimately fit relative to established therapies. But as a phase 1 result, it points to two things developers care about early: whether the target can be engaged deeply, and whether the dosing schedule can be practical. On both counts, the results appear supportive of more advanced testing.
Why ANGPTL3 is drawing attention
ANGPTL3 has become an important node in lipid research because it sits upstream of pathways that affect triglyceride metabolism and other atherogenic lipids. The genetic observation cited by the authors gives the target unusual appeal. When naturally occurring variants point in the same direction as a drug’s intended effect, developers gain a stronger biological rationale than they would from animal data alone.
That does not guarantee success. Many promising lipid-lowering approaches have struggled once tested in larger and more diverse populations, or when outcome studies examined whether biomarker improvements translated into fewer heart attacks or strokes. Even so, the zodasiran data strengthen the idea that RNA interference could become a practical way to modulate liver-produced proteins involved in cardiometabolic disease.
The dosing pattern is also notable. The main study used injections on days 1 and 29, and the extension moved to dosing every 12 weeks. If those schedules hold up in later trials, they could offer a different adherence profile from daily oral medicines or more frequently administered injectables.
What comes next
The authors conclude that the results indicate a favorable safety profile for zodasiran and support further studies in a wide spectrum of dyslipidemias. That is the correct level of restraint for phase 1. The trial was small, and it was designed to show early safety and pharmacodynamic activity rather than definitive clinical benefit. But the absence of serious treatment-related adverse events, along with the reported scale of ANGPTL3 and triglyceride reduction, gives the drug a credible case for continued development.
The immediate question is how zodasiran performs in larger controlled studies, especially in patients with persistent lipid abnormalities despite standard therapy. Later-stage trials will need to clarify how reproducible the lipid effects are, whether LDL-related benefits extend meaningfully across subgroups, and whether any safety issues emerge with broader exposure and longer follow-up.
For now, the phase 1 report positions zodasiran as one of the more interesting RNA-based entrants in cardiometabolic medicine. The promise is not just lower triglycerides, but a potentially versatile mechanism that could be used across multiple dyslipidemia phenotypes. Whether that early promise becomes a clinically important therapy will depend on the next rounds of evidence, but this first final report gives the field a solid reason to keep watching.
This article is based on reporting by Nature Medicine. Read the original article.
Originally published on nature.com