An oral follow-on option for a familiar obesity problem
One of the hardest parts of obesity treatment is not losing weight in the first place, but holding onto those gains once therapy changes or stops. A new phase 3b study in
Nature Medicine
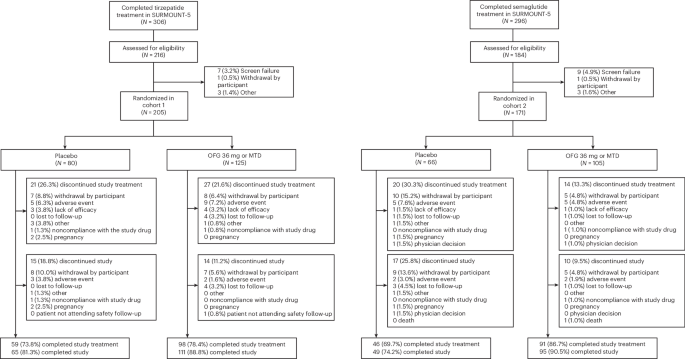
addresses that maintenance problem directly, testing whether a once-daily oral GLP-1 receptor agonist can help people preserve weight loss already achieved with injectable medicines.The drug is orforglipron, a nonpeptide oral GLP-1 receptor agonist. In the newly published ATTAIN-MAINTAIN trial, researchers enrolled people who had previously been treated with tirzepatide or semaglutide during the SURMOUNT-5 study. Participants were then randomized in a double-blind, placebo-controlled design to receive either daily oral orforglipron or placebo.
The core question was practical and clinically important: after patients reached a body-weight plateau on injectable therapy, could an oral medicine help them keep more of that reduction over the following year?
What the trial found
The answer, based on this study, was yes. Among participants in the tirzepatide-experienced cohort who had reached a weight plateau, those assigned to orforglipron maintained a model-based estimate of 74.7% of their body-weight reduction at week 52. The placebo group maintained 49.2%. That produced an estimated treatment difference of 25.5 percentage points, with a reported P value below 0.001.
Results were also strong in the semaglutide-experienced cohort. Participants on orforglipron maintained a model-based estimate of 79.3% of their weight reduction at week 52, compared with 37.6% for placebo. The estimated treatment difference was 41.7 percentage points, again with P < 0.001.
The paper says all key secondary endpoints were met as well, although the abstract does not detail each one individually. It also reports improvements in cardiometabolic risk factors and a safety profile generally similar to injectable GLP-1 receptor agonists.
The most common adverse events were gastrointestinal effects, described as mostly mild to moderate in severity. That detail matters because tolerability often determines whether a therapy can function outside tightly controlled trial settings.
Why this result matters
Obesity medicine has advanced rapidly, but many real-world questions now revolve around sequencing and persistence rather than only first-line efficacy. Injectable incretin-based drugs have produced meaningful weight loss for many patients, yet maintaining those benefits usually requires ongoing treatment. That creates challenges around access, adherence, patient preference, and long-term logistics.
The value proposition in this study is not that an oral drug newly outperformed a leading injectable during initial treatment. Instead, it is that an oral agent may offer a way to reduce weight regain after patients have already responded to injectable therapy. In other words, this is about the second phase of obesity care: maintenance.
The authors explicitly frame orforglipron as a potentially scalable option for minimizing weight changes after injectable therapy. That is an important phrase. Daily oral treatment could, in principle, fit more easily into some health systems and some patients’ routines than continued injections. The study does not prove that every patient would prefer that path, but it does support the idea that an oral maintenance strategy may be clinically credible.
For clinicians, the result adds a new decision point. If a patient has responded to tirzepatide or semaglutide but needs or wants a different long-term approach, an oral maintenance option could widen the treatment pathway. For payers and health systems, the question becomes whether maintenance therapy can preserve enough benefit to justify ongoing coverage in a cost-sensitive environment.
What the study does and does not show
The trial is promising, but its limitations are clearly stated. Most notably, there was no comparator arm in which patients simply continued on injectable obesity-management medication. That means the study cannot answer a central practical question: how does switching to oral orforglipron compare with just staying on the injectable drug that produced the original weight loss?
That missing comparison shapes how these results should be interpreted. The study shows that orforglipron outperformed placebo for maintenance. It does not show that switching to orforglipron is better than remaining on tirzepatide or semaglutide, or even equivalent to doing so.
The trial duration is another constraint. Participants were followed for one year. That is long enough to matter, but obesity is a chronic condition, and maintenance decisions often stretch across multiple years. Whether the relative benefit seen here persists, narrows, or changes over longer periods remains unanswered in this publication.
There is also the broader issue of implementation. Trial populations are selected and monitored in ways that routine care often is not. An oral therapy may appear simpler than injections, but real-world adherence to daily medication can still be uneven. The publication supports clinical potential, not guaranteed system-wide success.
A signal of where obesity treatment is headed
Even with those caveats, the study reflects a broader change in obesity care. The field is no longer focused only on whether weight can be reduced. Increasingly, it is asking how to manage transitions between therapies, how to sustain metabolic improvement, and how to make treatment durable at scale.
That is why this trial stands out. It addresses a problem that follows success. Patients who respond to modern injectable agents still need a long-term strategy. An oral maintenance option, if validated further and adopted in practice, could become part of that strategy rather than a niche alternative.
The study’s randomized, placebo-controlled design gives the findings weight, and the differences reported in both prior-treatment cohorts were substantial. The absence of a continued-injectable comparator keeps the result from being definitive on treatment sequencing, but it does not erase the importance of the signal.
For now, the most defensible takeaway is straightforward: in people who had already lost weight with tirzepatide or semaglutide and reached a plateau, daily oral orforglipron helped preserve more of that reduction over 52 weeks than placebo did. In a field increasingly concerned with long-term maintenance, that is meaningful progress.
The trial is registered at ClinicalTrials.gov under NCT06584916.
This article is based on reporting by Nature Medicine. Read the original article.
Originally published on nature.com