An Old Drug Gets a Modern Test
Digoxin has been part of cardiovascular medicine for generations, but its role in contemporary heart failure care has remained unsettled. A newly published randomized controlled trial in Nature Medicine puts that question back into focus with a large, placebo-controlled test of low-dose digoxin in patients with symptomatic chronic heart failure and a left ventricular ejection fraction of 50% or less.
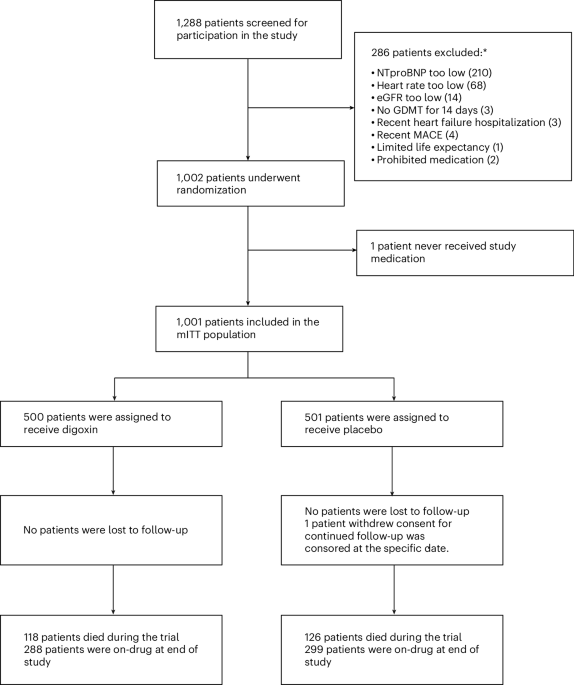
The study, known as the DECISION trial, enrolled 1,001 patients and randomized them to either low-dose digoxin or placebo. Investigators targeted serum digoxin concentrations of 0.5 to 0.9 ng/ml, a range meant to preserve potential benefit while limiting toxicity concerns that have long shadowed the drug. Participants had a mean age of 72 years, 28% were women and 29% had atrial fibrillation.
The topline result was mixed. Over a median follow-up of 36.5 months, the trial did not show a statistically significant reduction in its primary composite outcome: total worsening heart failure events, defined as total hospitalizations or urgent hospital visits for worsening heart failure, plus cardiovascular mortality. That means the study did not deliver the kind of clear positive result that would immediately reset guidelines or practice.
Still, the numbers did not point toward failure in a simple sense. Event counts were lower in the digoxin group than in the placebo group, suggesting a possible clinical signal that stopped short of conventional statistical significance.
What the Trial Found
In the digoxin group, 238 primary-outcome events occurred in 131 of 500 patients. In the placebo group, 291 primary-outcome events occurred in 152 of 501 patients. That translated to a rate ratio of 0.81, with a 95% confidence interval of 0.61 to 1.07 and a P value of 0.133.
On the narrower count of worsening heart failure events alone, the totals were 155 in the digoxin arm and 203 in the placebo arm, corresponding to a rate ratio of 0.76 with a 95% confidence interval of 0.54 to 1.05. Cardiovascular mortality was similar between groups: 83 patients, or 17%, in the digoxin group and 88 patients, or 18%, in the placebo group, with a hazard ratio of 0.93.
Those figures matter because they show the study was not negative across every measure in a directional sense. Instead, it produced a recurring pattern of somewhat fewer events in patients receiving digoxin, without crossing the statistical threshold for the primary endpoint. For clinicians and researchers, that puts the result into a familiar but consequential category: not definitive, but difficult to dismiss.
It also underscores why digoxin has remained controversial. The drug is inexpensive and widely known, and earlier studies suggested it might offer benefit, especially in reducing hospitalizations. Yet uncertainty about safety, the evolution of heart failure therapy and the lack of modern randomized evidence have kept it from regaining a secure place in standard regimens.