A gene-edited transplant approach reaches an early clinical milestone
A first-in-human study published in Nature Medicine reports early clinical results for a CRISPR-Cas9-edited allogeneic hematopoietic cell transplant designed for patients with high-risk acute myeloid leukemia and myelodysplastic syndrome. The treatment strategy uses donor cells that have had the CD33 target removed, allowing physicians to follow transplantation with the CD33-directed drug gemtuzumab ozogamicin without exposing normal donor-derived myeloid cells to the same level of risk.
The logic behind the approach is straightforward but technically ambitious. Patients with high-risk AML or MDS can relapse even after allogeneic hematopoietic cell transplantation. One challenge in post-transplant maintenance is that therapies intended to suppress or eliminate residual leukemia can also damage healthy donor cells. The investigational product in this trial, tremtelectogene empogeditemcel, or trem-cel, is intended to solve part of that problem by deleting CD33 from the donor graft before infusion.
What the phase 1/2a trial found
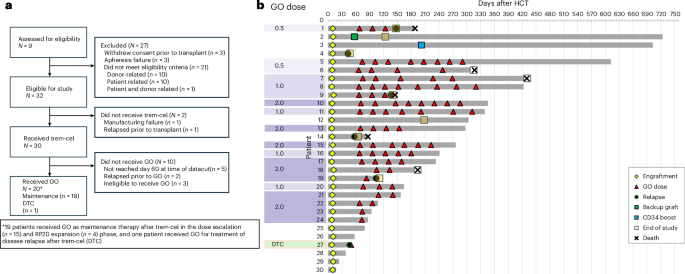
The multicenter, open-label phase 1/2a study enrolled adult patients with AML or MDS who were considered to have a high risk of relapse. After myeloablative conditioning, patients received trem-cel. Some participants then went on to receive maintenance gemtuzumab ozogamicin on 28-day cycles at doses ranging from 0.5 mg/m2 to 2.0 mg/m2.
The primary safety endpoint was neutrophil engraftment by day 28. According to the published report, all 30 patients who received trem-cel met that endpoint. Median time to neutrophil engraftment was 10 days, with a 95% confidence interval of 9 to 10 days. That matters because delayed or failed engraftment is one of the central risks in any transplant strategy, and particularly in one involving genome-edited donor cells.
Nineteen patients received post-transplant gemtuzumab ozogamicin maintenance, including 15 in the phase 1 dose-escalation portion and four in phase 2 dose expansion. The study reports that gemtuzumab ozogamicin was safely tolerated up to the recommended phase 2 dose of 2 mg/m2. The paper also notes that the trial was stopped early and serves as the final report including the completed phase 1 portion.
Why CD33 matters in this disease setting
CD33 is an established therapeutic target in myeloid malignancies, including AML, but it is also expressed on normal myeloid cells. That overlap is one reason CD33-targeted maintenance can be difficult after transplantation: the same antigen that marks leukemia cells can also mark the donor-derived blood-forming system physicians are trying to restore. By removing CD33 from donor hematopoietic cells before transplant, researchers aimed to create a graft that would remain functional while becoming less vulnerable to later CD33-directed treatment.
If that concept holds up in larger studies, it could point toward a broader model for transplant oncology: edit the donor graft first, then use targeted drugs more aggressively after transplant. In other words, the cell therapy is not meant only to replace diseased marrow. It is also meant to reshape what maintenance treatment becomes possible afterward.
What is encouraging and what remains unresolved
The most encouraging part of the report is that the edited graft appears to have cleared a basic but essential clinical hurdle. Universal day-28 neutrophil engraftment across 30 treated patients is a strong signal that the edited product can reconstitute myeloid recovery on the timeline clinicians expect. The tolerance of maintenance gemtuzumab ozogamicin up to the recommended phase 2 dose adds further support to the biological premise of protecting donor cells from on-target toxicity.
At the same time, this is still an early-stage study with a limited patient count. The article summary references additional secondary endpoints including graft-versus-host disease, graft failure, transplant-related mortality, the percentage of CD33-negative myeloid cells, and survival, but the supplied source text provides only a partial view of those outcomes. That means the most defensible takeaway is not that the strategy has solved relapse risk, but that it has demonstrated feasibility and early safety signals in a setting where both are hard won.
A notable step for gene editing in transplantation
Gene editing in blood disorders has often been framed around autologous cell therapies, where a patient’s own cells are modified and returned. This trial instead centers on an allogeneic product intended for transplant recipients with aggressive disease. That makes it notable beyond AML alone. It suggests CRISPR-based editing may have a role not only in correcting inherited defects, but in engineering donor grafts to function as more resilient therapeutic platforms.
For now, the study reads as a proof-of-concept milestone rather than a practice-changing result. But in high-risk AML and MDS, where relapse after transplant remains one of the field’s hardest problems, a transplant product deliberately built to withstand targeted maintenance therapy is a serious idea. The early clinical record published this week suggests that idea has moved beyond theory and into real, if still preliminary, patient experience.
This article is based on reporting by Nature Medicine. Read the original article.
Originally published on nature.com