Replimune’s second FDA rejection keeps pressure on biotech regulation and cancer drug risk
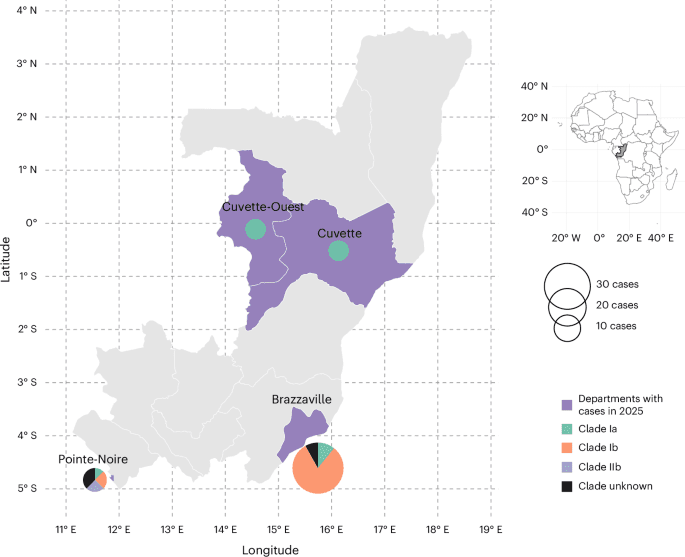
Replimune’s skin cancer therapy has reportedly been rejected by the FDA again, extending a high-profile regulatory setback that had already become a flashpoint in agency debates.
- Replimune’s skin cancer drug was reportedly rejected by the FDA for a second time.
- The case had already become a flashpoint in FDA debates.