A late-stage result for a difficult rare disease
Benralizumab significantly reduced the risk of disease flare in patients with FIP1L1::PDGFRA-negative hypereosinophilic syndrome, according to a phase 3 trial published in Nature Medicine. The result is notable for a rare disorder with heterogeneous symptoms, frequent diagnostic delays, and limited treatment options beyond steroids and disease-specific therapies that do not apply to every patient subgroup.
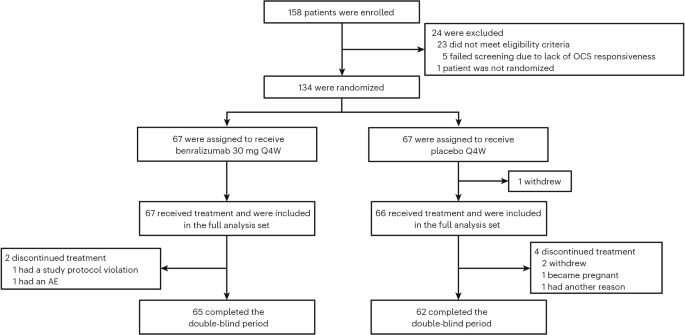
The randomized, double-blind, placebo-controlled NATRON study enrolled 133 patients, who received either benralizumab 30 mg every four weeks or placebo for 24 weeks on top of background therapy. The primary endpoint was time to first hypereosinophilic syndrome flare. Investigators reported a hazard ratio of 0.35 for benralizumab versus placebo, with a 95% confidence interval of 0.18 to 0.69 and a P value of 0.0024.
In practical terms, that means the treated group experienced a substantially lower risk of flare during the study period. For a disease defined by eosinophil-driven damage across multiple organ systems, that reduction could matter clinically even before longer-term outcomes are fully characterized.
What hypereosinophilic syndrome is
Hypereosinophilic syndrome, or HES, refers to a group of conditions marked by persistently elevated eosinophils together with evidence that those immune cells are causing tissue injury. The syndrome can affect multiple organs, and the presentation can vary widely from patient to patient. That variability contributes to delayed diagnosis and makes treatment planning difficult.
Some patients have molecular abnormalities that point to highly effective targeted therapies. The article notes that people with the FIP1L1::PDGFRA fusion gene can respond remarkably well to imatinib. But those options do not cover patients who are negative for that rearrangement. The NATRON trial focused specifically on that unmet-need population.
Why benralizumab was a logical candidate
Benralizumab is an anti-IL-5 receptor alpha antibody designed to deplete eosinophils. It already has an established role in severe eosinophilic asthma and eosinophilic granulomatosis with polyangiitis. Its mechanism made it a plausible candidate for HES, where eosinophils are directly implicated in disease activity.
The new phase 3 data move the drug from promise to stronger clinical evidence in this setting. Earlier signs of benefit are important in rare diseases, but regulators and clinicians generally look for placebo-controlled late-stage trials before changing routine practice. That is what gives this study weight.
Safety profile remained in line with expectations
Safety will remain central in any add-on treatment for a chronic, relapsing disease. In the trial, adverse events occurred in 64.2% of benralizumab-treated patients and 66.7% of those on placebo. The researchers said the drug’s safety findings were consistent with its known profile.
That does not mean safety questions are closed. Twenty-four weeks is enough to assess short-term tolerability and support a primary efficacy readout, but it is not the same as years of follow-up across broader real-world populations. Even so, the phase 3 result appears encouraging because it pairs a meaningful flare reduction with no obvious signal of markedly higher adverse-event burden than placebo in the study window.
What the result could change
For clinicians, the study provides stronger evidence for an eosinophil-targeted strategy in FIP1L1::PDGFRA-negative HES. For patients, it points to the possibility of reducing flare frequency without relying solely on treatments that may have substantial long-term side effects or limited disease specificity.
Because HES is rare and clinically diverse, treatment standards can lag behind those for more common inflammatory diseases. Large, clean trials are harder to run, and patient subgroups can behave differently. That makes a statistically significant placebo-controlled phase 3 result especially consequential. It gives physicians a firmer basis for discussing risk, benefit, and sequencing in treatment decisions.
It may also influence future research design. If eosinophil depletion continues to look effective, investigators may explore durability of response, steroid-sparing potential, subgroup effects, and use in combination or earlier-line settings. Real-world studies will be important as well, particularly in patients whose disease courses are more complex than those typically seen in tightly controlled trials.
A meaningful step, not the final word
The NATRON data do not eliminate the challenges of HES. The syndrome remains biologically diverse, and even a positive trial in one molecularly defined population will not answer every clinical question. But it does represent one of the stronger treatment signals yet for patients who lack the best-known targetable rearrangement.
That matters because rare-disease progress often arrives in increments: a better endpoint here, a clearer subgroup there, a trial robust enough to change expert confidence. Benralizumab’s phase 3 performance appears to be one of those increments. It suggests that a drug already used to control eosinophil-driven inflammation elsewhere may have a broader role in reducing damaging flares in HES, with a safety profile that did not depart from expectations during the trial period.
For patients and physicians dealing with a rare condition that is easy to miss and hard to manage, that is a meaningful development.
This article is based on reporting by Nature Medicine. Read the original article.
Originally published on nature.com