A shift from retrospective review to live oversight
The US Food and Drug Administration is launching a pilot program to collect and review clinical trial data in real time, an approach that could change how drug studies are monitored and how regulators interact with sponsors before formal submissions are complete.
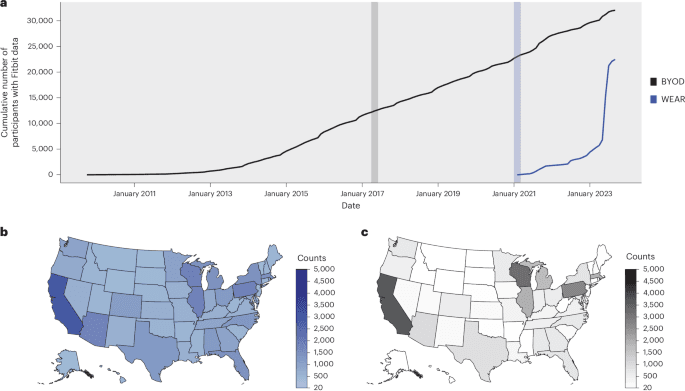
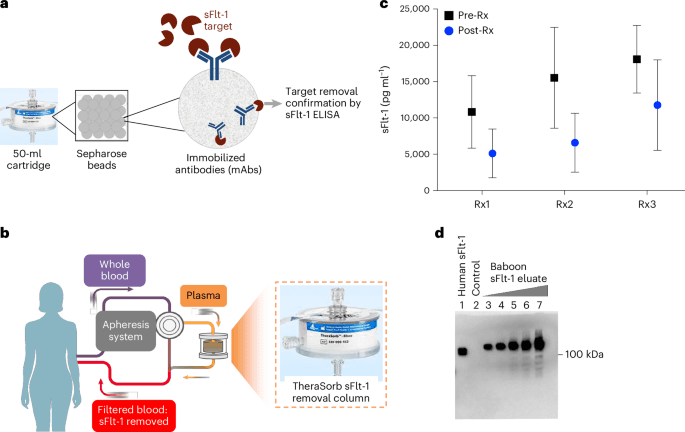
According to the agency announcement reported on April 28, scientific reviewers will be able to access trial data as it flows in. AstraZeneca and Amgen are serving as the initial test cases. That alone makes the program significant. Rather than waiting for a finished package of results at the end of a study cycle, the FDA is experimenting with a more continuous model of regulatory visibility.
The move reflects a broader modernization push in life sciences and regulation. Clinical research has become increasingly digital, global, and data-rich, but regulatory review has often remained anchored to batch-style submissions assembled after the fact. Real-time access would not eliminate the need for final analysis, but it could compress the distance between evidence generation and regulatory understanding.
What real-time review could change
If implemented effectively, the pilot could affect several parts of the drug development process. Earlier visibility may allow FDA reviewers to spot data quality issues, inconsistencies, or operational problems sooner. It could also create more structured dialogue between companies and regulators while studies are underway rather than after problems have already become expensive or irreversible.
In principle, real-time review could improve:
- Detection of data anomalies before they propagate through a larger trial program
- Clarity around endpoints, protocol adherence, and study conduct
- Regulatory preparedness ahead of eventual filing decisions
- Efficiency in the final review process if core questions are surfaced earlier
That does not mean the agency is moving to approve drugs on partial evidence. Final evidentiary standards remain crucial. But earlier access to the underlying stream of information may reduce surprises and help regulators build context while a study is still active.