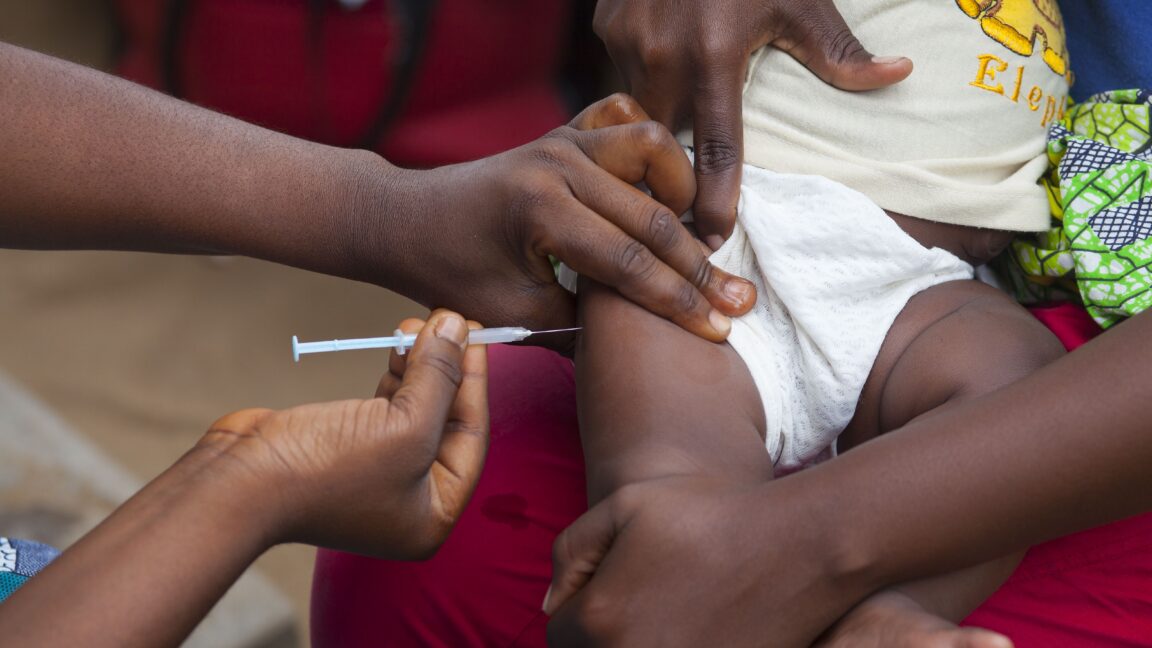
WHO Issues Rare Public Condemnation
The World Health Organization has publicly condemned a US-funded clinical trial planned for Guinea-Bissau as "unethical," marking a sharp rebuke of research backed by the Centers for Disease Control and Prevention. WHO Director-General Tedros Adhanom Ghebreyesus stated during a news conference: "Of course, a sovereign country can decide whatever they want, but as far as WHO's position is concerned, it's unethical to proceed with this study."
The randomized controlled trial, partially funded by a $1.6 million CDC grant, would study the general health effects of giving the hepatitis B vaccine at birth to approximately 14,500 infants in the West African nation. Half would receive the birth dose, while the other half would receive their first dose at two months, the current standard of care in Guinea-Bissau.
Why WHO Considers It Unethical
The WHO's objections center on the study's design and context. The hepatitis B birth dose vaccine has been used for over three decades and is included in the national immunization schedules of more than 115 countries. Guinea-Bissau has one of the world's highest hepatitis B infection rates, with approximately one in five residents carrying the virus. An estimated 90% of newborns infected during childbirth become chronic carriers at high risk for cirrhosis and liver cancer.
WHO stated that "exploiting scarcity is not ethical: resource constraints cannot be used to justify withholding proven care in a research study involving people." The organization also noted the study protocol does not ensure basic harm reduction measures, such as screening pregnant women and vaccinating exposed newborns.
Trial Suspended Amid Controversy
Guinea-Bissau's Minister of Public Health, Quinhin Nantote, announced the study's suspension pending review. The Africa CDC is sending a team to help the country evaluate the research. The trial, led by the Bandim research group, was awarded a non-competitive, unsolicited CDC grant.
The controversy has drawn comparisons to historical ethical failures. Vaccine developer Paul Offit called the trial "RFK Jr.'s Tuskegee Experiment," referencing the infamous syphilis study. The trial's timing coincides with a federal advisory panel, selected by HHS Secretary Robert F. Kennedy Jr., voting to stop recommending hepatitis B shots for all newborns in the United States.
Competing Claims
An HHS spokesperson defended the trial as based on "the highest scientific and ethical standards." However, following initial contradictions about the study's status, HHS ultimately acknowledged the trial had been paused pending further evaluation. Guinea-Bissau had formally decided in 2024 to add the hepatitis B birth dose to its national schedule, with implementation planned by 2028.
This article is based on reporting by Ars Technica. Read the original article.
Originally published on arstechnica.com