A closer look at semaglutide in a higher-risk subgroup
A new prespecified analysis from the SELECT randomized trial suggests semaglutide may provide cardiovascular benefit in patients who are not only living with obesity and atherosclerotic cardiovascular disease, but who also appear to be at elevated risk for substantial liver fibrosis. The analysis, published in Nature Medicine, extends interest in semaglutide beyond weight management and broad cardiovascular risk reduction into a clinically important subgroup shaped by liver disease risk.
In the main SELECT trial, once-weekly subcutaneous semaglutide reduced major adverse cardiovascular events, or MACE, by 20% versus placebo in patients with obesity and established atherosclerotic cardiovascular disease who did not have diabetes. The new analysis asked whether that benefit also held among patients considered at higher risk of liver fibrosis, a population of growing importance as metabolic dysfunction-associated steatotic liver disease becomes more common.
What the analysis found
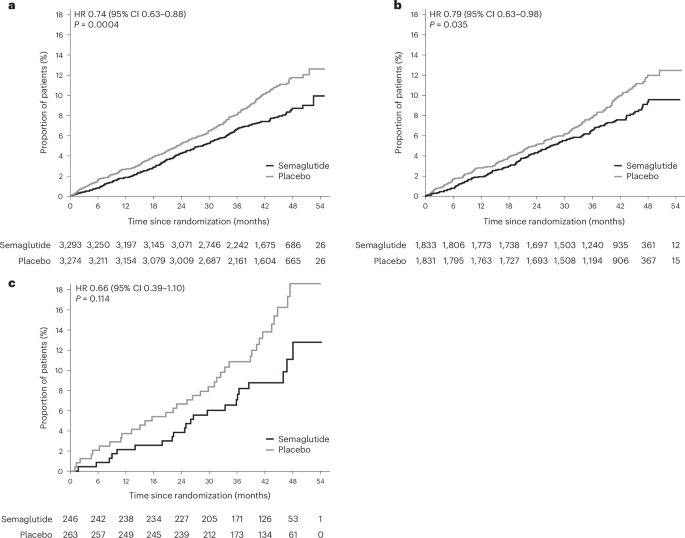
Investigators assessed liver biochemical tests and steatosis risk over 104 weeks and examined cardiovascular outcomes in subgroups defined by Fibrosis-4, or FIB-4, scores. In patients meeting one of the fibrosis-risk thresholds, semaglutide was associated with lower rates of major adverse cardiovascular events compared with placebo.
The supplied text reports the following subgroup results:
- In patients with baseline FIB-4 scores of at least 1.3, MACE was reduced by 26%, with a hazard ratio of 0.74 and a P value of 0.0004.
- Using age-specific thresholds of at least 1.3 for those younger than 65 years and at least 2.0 for those 65 and older, MACE was reduced by 21%, with a hazard ratio of 0.79 and a P value of 0.035.
- In patients with FIB-4 greater than 2.67 at any age, MACE was reduced by 34%, with a hazard ratio of 0.66, though the reported P value of 0.11 indicates that result did not reach conventional statistical significance.
Researchers also reported a 28% greater decrease in fatty liver index with semaglutide versus placebo, with a highly significant P value below 0.0001.
Why liver fibrosis risk matters here
The analysis sits at the intersection of cardiometabolic disease and liver disease. The paper notes that overweight and obesity are established risk factors for metabolic dysfunction-associated steatotic liver disease, or MASLD. As fat accumulates in the liver, some patients progress from steatosis to metabolic dysfunction-associated steatohepatitis and eventually to advanced fibrosis.
That overlap is one reason the subgroup is so important. Patients with obesity and cardiovascular disease may also face underrecognized liver risk, and clinicians increasingly want to know whether therapies can address both domains. The new results do not show that semaglutide reverses fibrosis directly, but they do indicate that the drug’s cardiovascular benefit remains present in people screened as being at increased fibrosis risk, while liver-related risk markers also move in a favorable direction.
How to interpret the findings
Because this was a prespecified secondary analysis rather than the trial’s primary endpoint population definition, the findings are best read as an important extension of SELECT rather than a standalone verdict on liver disease treatment. The primary takeaway from the supplied text is not that semaglutide has been newly proven as a fibrosis therapy. Instead, it is that semaglutide continued to reduce cardiovascular events in patients at elevated risk for substantial liver fibrosis, broadly consistent with the overall SELECT population.
That nuance matters. FIB-4 and fatty liver index are useful clinical tools, but they are not the same as biopsy-confirmed fibrosis outcomes. Even so, prespecified analyses carry more weight than purely retrospective fishing expeditions, and these data will likely add momentum to the broader view of semaglutide as a cardiometabolic drug with clinically relevant effects beyond weight loss alone.
Why this could influence practice and research
For physicians, the analysis reinforces the need to think across specialties. Patients with obesity, cardiovascular disease, and signs of liver risk often sit between cardiology, endocrinology, hepatology, and primary care. Evidence that one therapy may improve outcomes in overlapping risk domains is therefore highly relevant to everyday treatment decisions.
For researchers and drug developers, the results strengthen the case for continued study of GLP-1-based therapies in populations defined by liver disease risk. They also underscore how trials built around cardiovascular endpoints can yield insights into broader metabolic disease patterns when investigators plan secondary analyses in advance.
The conclusion offered in the paper is measured: semaglutide reduced major adverse cardiovascular events versus placebo in patients at risk for substantial liver fibrosis, similar to what was seen in the overall SELECT trial population. That is not the final word on semaglutide and liver disease, but it is a meaningful signal in a patient group with large unmet need.
This article is based on reporting by Nature Medicine. Read the original article.
Originally published on nature.com