A first clinical test of a disease-specific approach
Preeclampsia remains one of the most dangerous complications of pregnancy, particularly when it emerges early enough to force delivery far before term. In a pilot trial published in Nature Medicine, researchers tested an experimental strategy designed to lower levels of soluble Fms-like tyrosine kinase 1, or sFlt-1, a placental protein widely implicated in the disease process. The study does not establish a new standard of care, but it offers an early sign that a targeted, mechanism-based intervention may be possible in a condition where treatment options have long been limited.
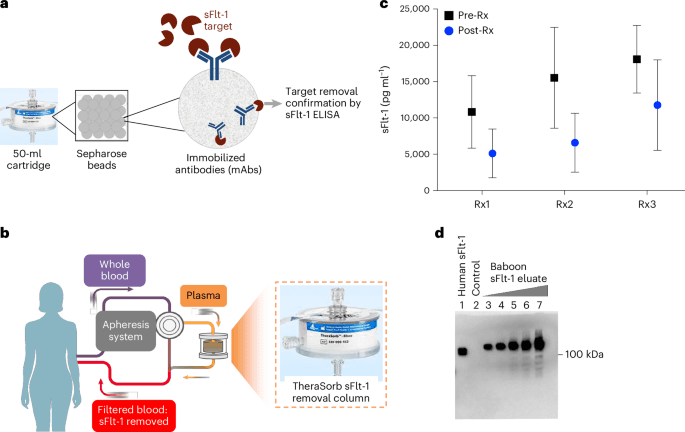
The investigators developed an antibody-based apheresis approach intended to selectively remove circulating sFlt-1 from the mother’s blood. Their report describes preclinical work in pregnant baboons and an open-label, single-arm human study in women with preterm and very preterm preeclampsia. The trial’s primary focus was not proof of efficacy, but safety and tolerability.
Why sFlt-1 matters
The rationale for the study is straightforward. The paper states that sFlt-1 plays a central role in the pathogenesis of preeclampsia. That makes it an unusually attractive therapeutic target: not just a marker associated with the condition, but a factor believed to help drive it. In preeclampsia, clinicians often have to balance maternal risk against fetal immaturity, because there is no disease-specific treatment that reliably halts the underlying process.
A therapy that could reduce the biological pressure of the disease, even temporarily, could matter most in pregnancies where every additional day in utero has clinical value. The authors therefore focused on women with very preterm disease, where prolonging pregnancy, stabilizing maternal status, or both could be meaningful.