A Preventive Approach to Hereditary Cancer
A new study published in Nature Medicine has revealed promising early results for a neoantigen vaccine designed to prevent cancer in individuals with Lynch syndrome, one of the most common hereditary cancer predispositions. The vaccine, described as an "off-the-shelf" formulation that does not require customization for each patient, showed both safety and the ability to trigger meaningful immune responses against the molecular markers associated with Lynch syndrome cancers.
The findings represent a conceptual shift in cancer treatment. While therapeutic cancer vaccines — designed to treat existing tumors — have been an active area of research for decades with mixed results, preventive cancer vaccines aim to prime the immune system to recognize and destroy cancer cells before they form detectable tumors. If this approach proves effective in larger trials, it could fundamentally change how medicine addresses hereditary cancer risk.
Understanding Lynch Syndrome
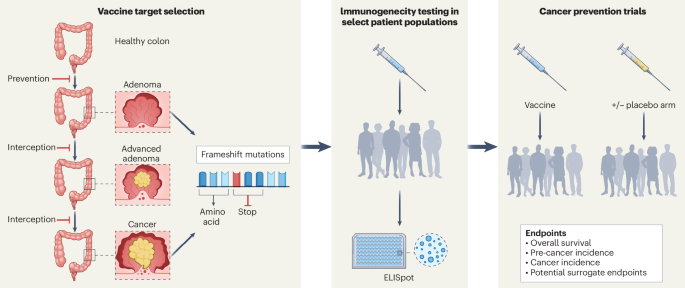
Lynch syndrome is a genetic condition caused by mutations in DNA mismatch repair genes. These genes normally function as a proofreading system for DNA replication, catching and correcting errors that occur when cells divide. When mismatch repair is impaired, mutations accumulate at an accelerated rate, dramatically increasing the risk of developing several types of cancer — most notably colorectal cancer, endometrial cancer, and cancers of the stomach, ovary, and urinary tract.
Approximately one in every 280 people carries a Lynch syndrome mutation, making it one of the most prevalent hereditary cancer syndromes. Individuals with the condition face a lifetime colorectal cancer risk of 40 to 80 percent, compared to roughly 4 percent in the general population. Current management relies on intensive surveillance through frequent colonoscopies and other screening procedures, along with prophylactic surgeries in some cases. A vaccine that could reduce cancer incidence in this population would represent a transformative advance.
How the Neoantigen Vaccine Works
The vaccine targets neoantigens — novel proteins that appear on the surface of cells with mismatch repair deficiency. Because Lynch syndrome cancers accumulate mutations in predictable patterns, the resulting neoantigens are shared across many patients rather than being unique to each individual. This shared neoantigen profile is what makes an off-the-shelf vaccine feasible, in contrast to the personalized neoantigen vaccines that require sequencing each patient's tumor and manufacturing a custom formulation.
The vaccine is designed to train the immune system's T cells to recognize these shared neoantigens and mount an attack against any cells displaying them. In theory, this would create an immune surveillance system that could detect and eliminate mismatch repair-deficient cells at the earliest stages — potentially before they evolve into clinically detectable cancers. The approach essentially weaponizes the immune system against the very molecular signature that defines Lynch syndrome cancers.
Trial Results and Safety Profile
The clinical trial enrolled individuals diagnosed with Lynch syndrome who had not yet developed cancer. Participants received the vaccine and were monitored for both safety and immune response. The study reported that the vaccine was well-tolerated, with no serious adverse events attributed to the treatment. Side effects were generally mild and consistent with those seen with other vaccine platforms, including injection site reactions and transient flu-like symptoms.
More importantly, the vaccine generated measurable immune responses in the trial participants. Researchers detected increased activity of T cells specifically targeting the Lynch syndrome-associated neoantigens, suggesting that the vaccine successfully primed the immune system to recognize the molecular markers of mismatch repair-deficient cells. The strength and durability of these immune responses will be critical factors in determining whether the vaccine can provide long-term protection against cancer development.
From Treatment to Prevention
The concept of cancer prevention vaccines has been proven in principle by the human papillomavirus (HPV) vaccine, which prevents cervical and several other cancers by blocking the virus that causes them. However, the Lynch syndrome vaccine represents a more ambitious application: preventing cancers that arise from inherited genetic mutations rather than from an infectious agent. There is no pathogen to block — instead, the vaccine must train the immune system to police the body's own mutating cells.
This approach could potentially be extended to other hereditary cancer syndromes, including BRCA1/2 mutations associated with breast and ovarian cancer, and Li-Fraumeni syndrome, which predisposes individuals to a wide range of cancers. Each syndrome produces characteristic patterns of mutations and neoantigens that could, in theory, be targeted by similar vaccine strategies. The Lynch syndrome trial serves as a proof of concept for an entirely new category of cancer prevention.
The Road Ahead
The early-stage results are encouraging but preliminary. Larger, longer-term trials will be needed to determine whether the immune responses generated by the vaccine actually translate into reduced cancer incidence. Cancer prevention studies inherently require long follow-up periods, since the endpoint — whether participants develop cancer — may take years or decades to assess. Researchers will also need to optimize dosing schedules, determine whether booster shots are needed, and identify which patient subgroups benefit most from vaccination.
Despite these caveats, the study represents a meaningful step toward a future where hereditary cancer risk is managed not through anxious surveillance and prophylactic surgery but through immunization. For the millions of people worldwide who carry Lynch syndrome mutations and live with the knowledge that cancer is a statistical probability rather than a remote possibility, a preventive vaccine offers something that screening alone cannot: the prospect of actually reducing the risk rather than merely catching the disease early.
This article is based on reporting by Nature Medicine. Read the original article.
Originally published on nature.com