The Cell's Grand Central Station
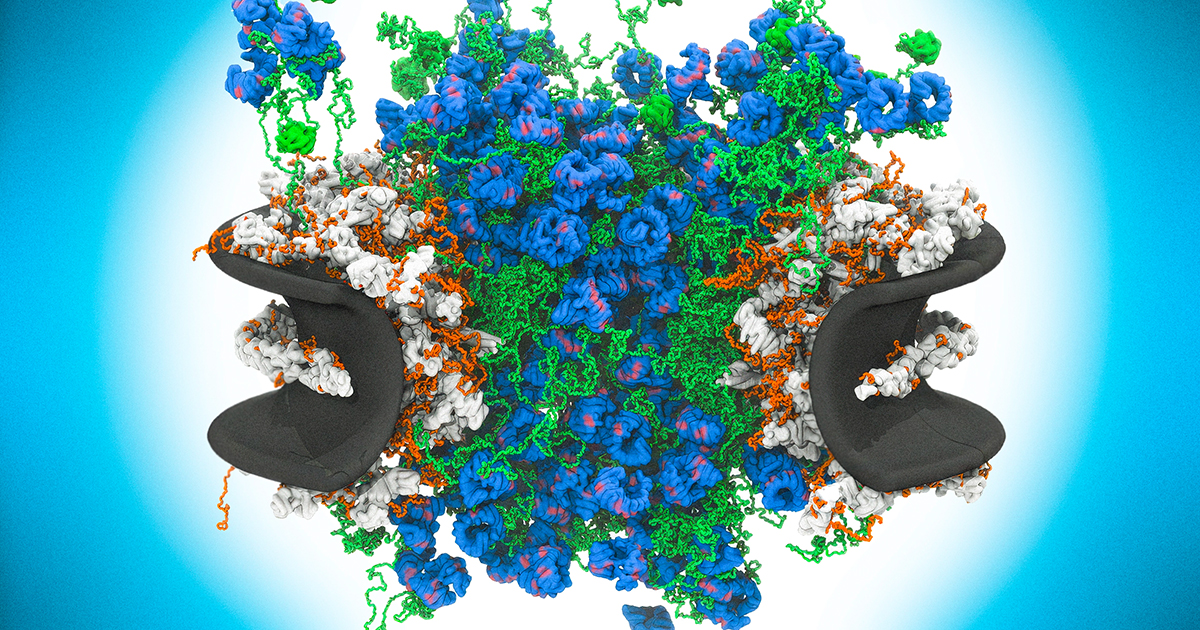
Every second, hundreds to thousands of molecules shuttle through nuclear pore complexes embedded in the membranes of your cells. These massive molecular machines — each composed of roughly 1,000 protein subunits arranged in an octagonal ring — serve as the sole gatekeepers between the cell's nucleus, where DNA is stored, and the cytoplasm, where proteins carry out their functions. Now, new high-resolution imaging is revealing a counterintuitive truth about how these essential structures work: their function depends on disorder.
The nuclear pore complex, or NPC, is one of the largest and most complex molecular assemblies in biology. At approximately 120 megadaltons in size, each pore dwarfs most cellular structures. A typical human cell contains several thousand of them, and together they regulate the bidirectional flow of messenger RNA, proteins, and signaling molecules that keeps the cell alive.
The Disordered Heart of the Machine
For decades, scientists assumed that the NPC's remarkable selectivity — allowing some molecules to pass freely while blocking others — must derive from a precisely organized internal structure. The expectation was that the pore's interior would prove to be a finely engineered tunnel with specific binding sites and mechanical gates.
Instead, researchers have discovered that the central channel of the nuclear pore is filled with a tangled mesh of intrinsically disordered proteins called FG-nucleoporins. These proteins, named for their repeated phenylalanine-glycine motifs, lack the well-defined three-dimensional structures that characterize most functional proteins. They wave and fluctuate constantly, forming a dynamic, gel-like barrier rather than a rigid gate.
This disordered mesh is what gives the NPC its selectivity. Small molecules and ions can diffuse through the gaps in the mesh, while larger molecules are blocked unless they carry a nuclear localization signal — a molecular passport recognized by transport receptor proteins. These receptors interact with the FG-nucleoporins through transient, weak binding events, essentially dissolving their way through the barrier.
New Imaging Illuminates the Dance
The latest advances in cryo-electron tomography and single-molecule fluorescence microscopy have provided unprecedented views of the NPC in action. Researchers can now watch individual transport events in real time, tracking how molecules approach the pore, enter the disordered mesh, navigate through the channel, and emerge on the other side.
These observations reveal that transport through the NPC is remarkably fast — a single translocation event can occur in as little as five to ten milliseconds — and highly efficient, with the pore handling hundreds of transport events per second. The disordered FG-nucleoporins facilitate this speed by providing a low-friction pathway for cargo-receptor complexes, which slide through the mesh without getting permanently stuck.
Why Disorder Matters
The NPC challenges a central dogma in molecular biology: that protein function requires defined structure. While many proteins fold into precise shapes that determine their activity, the FG-nucleoporins demonstrate that controlled disorder can be equally functional. Their flexibility allows the pore to accommodate cargo molecules of widely varying sizes, from small signaling proteins to massive ribosomal subunits, without requiring mechanical reconfiguration.
This adaptability would be impossible with a rigid gate mechanism. The disordered mesh acts more like a smart filter that responds dynamically to whatever is passing through it, expanding and contracting around cargo of different dimensions while maintaining its selective barrier function.
Implications Beyond Cell Biology
Understanding how the NPC uses disorder to achieve selective transport has implications beyond basic biology. Engineers designing nanoscale filtration systems and selective membranes are studying the NPC as inspiration for synthetic systems that can sort molecules by size and chemical properties without active energy input.
In medicine, mutations in nuclear pore components have been linked to neurodegenerative diseases, certain cancers, and premature aging syndromes. Detailed knowledge of how the NPC functions — and malfunctions — could open new therapeutic avenues for these conditions.
The nuclear pore complex stands as a powerful reminder that nature's most sophisticated machines do not always rely on precision engineering. Sometimes, the most effective solution is a carefully calibrated mess.
This article is based on reporting by Quanta Magazine. Read the original article.
Originally published on quantamagazine.org